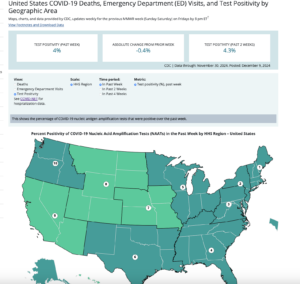
Moderna has begun clinical trials for a new flu vaccine that will use the same mRNA technology that was in the Covid vaccine. They will trial this drug on 50 people.
Fifty is not a very impressive number for a clinical trial but it doesn’t matter to the UK government, which has gone full-steam ahead with an order for at least 250 million vaccines per year from Moderna. The deal includes the construction of a new vaccine factory.
Moderna’s clinical trials in the U.S. will follow these 50 participants for up to 1 year but not longer than that.
Question: Does it feel like they are purposely not making these studies robust? See Dr. John Campbell’s take on the matter here.